Translational research is the direct link between basic research and clinical research:
scientists and doctors working together for the benefit of patients.
Knowledge transfer between researchers and doctors
Translational research is the adaptation of research results from the laboratory to the bedside. It accelerates the translation of scientific discoveries into medical applications for patients.
To achieve this, translational research requires a high level of collaboration between researchers and physicians to allow a two-way flow of knowledge. There is no longer a team of researchers on one side and a team of doctors on the other, but a multidisciplinary team of researchers and clinicians working closely together.
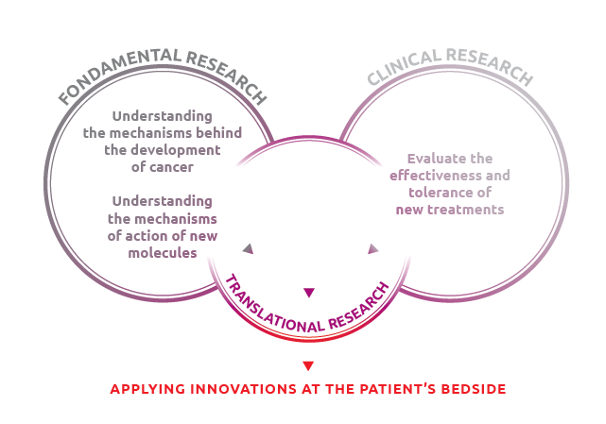
The objectives of translational research
- Develop clinical applications from a research discovery
- Encourage the exploration of new avenues, theories or concepts based on a clinical observation

The main objective of translational research is to accelerate the application of scientific discoveries so that patients can benefit from new health products as quickly as possible in clinical practice
Jean-Yves Blay – Président d’Unicancer
Sample collection
Translational research may involve the collection of clinical samples (tumour biopsies, blood samples, saliva samples, etc.) from patients participating in clinical trials.
Like all valuable resources, these samples are stored in “banks” under optimal conditions for their preservation and identification. These are known as tissue or biological sample banks.
These samples can then be made available to multidisciplinary teams after a research project has been submitted and after the research project has been evaluated: scientific and medical data, estimation of the expected benefit for patients at the end of the research.
Protection of patients’ personal data
Data protection is guaranteed throughout the process. Thus, the personal and health information of patients who have given their consent to the use of their biological samples for further research to advance knowledge and discoveries about their disease and future treatments is never disclosed to the managers of the biological sample banks or to the teams using the samples for their research projects.
Allowing the use of your samples, an investment for the future
The decision by patients to consent to the collection and subsequent use of their biological samples for research to advance knowledge of their disease and discoveries of future treatments is purely altruistic. Indeed, in the case of samples collected during clinical trials, the results of translational research are known years after the end of the clinical trials and will benefit other patients who will develop the same disease in the future, and rarely the patients who agreed to donate biological samples. This is particularly true in the case of cancer patients.


