- RWD

PEACE
- Prostate
PEACE: European Prostate Cancer Consortium
Prostate cancer: the second most common cancer in men with a steady increase over the years.
In 2020, according to the International Agency for Research on Cancer (IARC), 500,000 new cases of prostate cancer were reported in Europe, with the incidence varying from country to country. This European heterogeneity probably reflects the availability of tests to detect prostate cancer at an early stage.
Over the last two decades, phase III clinical trials have led to major changes in clinical practice by improving the treatment of prostate cancer which, together with earlier diagnosis, have probably contributed to the overall downward trend in mortality in Europe.
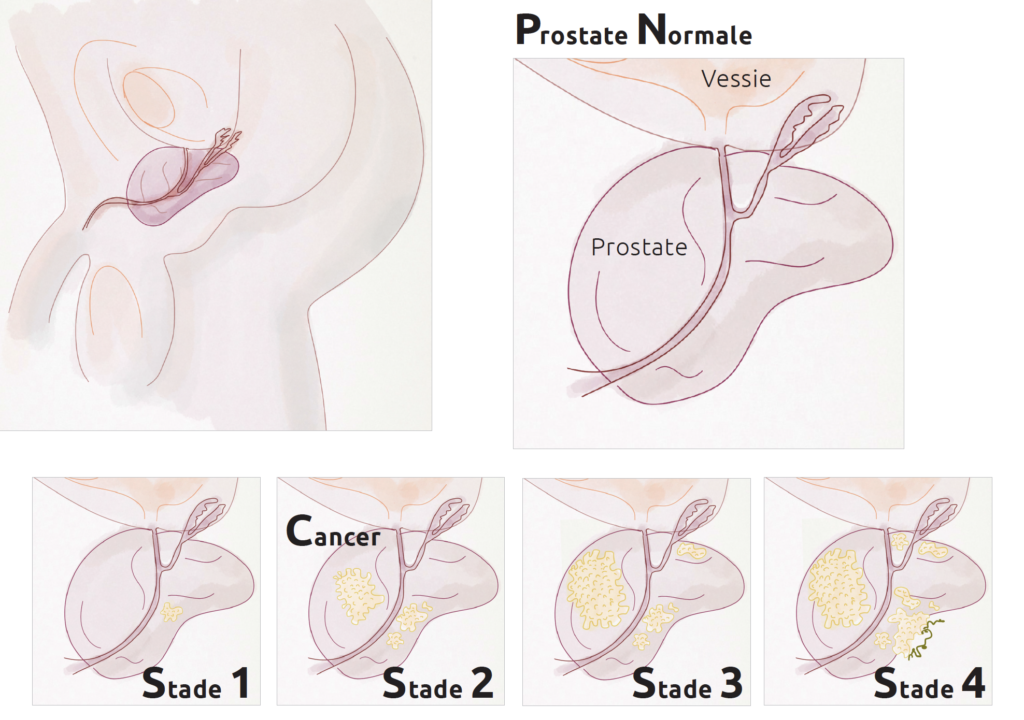
Stages of prostate cancer
The PEACE programme: a European grouping for the benefit of research
Numerous phase III trials have been conducted, providing patients with increasingly effective treatments. However, new studies now require larger and larger numbers of participating patients to further improve the prognosis for men at all stages of prostate cancer.
By coming together in national and European organisations, European genitourinary cancer specialists have helped to fill the ‘numbers gap’ in phase III clinical trials and are making a significant contribution to research and best practice in genitourinary cancer treatment. However, few of these organisations focus solely on prostate cancer.
In order to fill this gap, the Prostate Cancer Consortium in Europe (PEACE) was established in 2013. It is a pan-European research group open to all prostate cancer specialists practising in EU countries.
The PEACE consortium was set up to conduct comparative trials – specifically phase III studies – in Europe to evaluate new treatments for prostate cancer patients.
Since its inception, PEACE has set up several phase III trials to answer questions about the use of new treatment options for prostate cancer patients.
PEACE-1 (NCT01957436 ; EudraCT 2012-000142-35) ocuses on androgen deprivation therapy with or without local radiotherapy, with or without abiraterone acetate and prednisone, in patients with metastatic hormone-dependent prostate cancer
PEACE-2 (NCT01952223 ; EudraCT 2012-000566-38) is investigating cabazitaxel and pelvic lymph node directed radiotherapy in patients with localised prostate cancer and at least two high-risk features for relapse. It is planned to recruit 1048 patients. The duration of treatment will be three years and each patient will be followed for 10 years after completion of treatment.
PEACE-3 (NCT02194842 ; EudraCT 2014-001787-36)
PEACE-4 (NCT03819101 ; EudraCT 2017-004639-35)
PEACE-5 (NCT03569241) is a trial coordinated in Belgium by Piet Ost which is addressing the question of radiotherapy of metastases and is in the process of inclusion*.
PEACE-6 is a multi patient-group trial:
- PEACE-6-Oligo (PRESTO; NCT04115007) is a trial coordinated by Pierre Blanchard. This trial is in the process of being included and raises the question of radiotherapy of bone metastases
- PEACE-6-Unfit (NCT04916613; EudraCT 2020-003663-26) addresses the issue of intensifying treatment of metastatic and frail patients and is expected to begin enrolment in early 2022
The PEACE-7 study is being written and funded for high-risk prostate cancer patients